Is it tumor-induced osteomalacia (TIO)?
Connect the symptoms.

WHAT IS TIO?
TIO is a rare,
acquired form of hypophosphatemia1
TIO is typically caused by benign phosphaturic mesenchymal tumors that produce excess fibroblast growth factor 23 (FGF23) hormone.2 These tumors are1:
- Generally small and difficult to locate
- Located anywhere in the body, including within soft tissue and bone
WHY IS TIO OFTEN MISDIAGNOSED?
Symptoms of TIO are nonspecific, which may lead to misdiagnosis1
In a retrospective study, 95% (137/144) of patients were initially misdiagnosed3
Conditions that TIO is commonly misdiagnosed as include3,4:
- Intervertebral disc herniation
- Spondyloarthritis (including ankylosing spondylitis)
- Osteoporosis
- Rheumatoid arthritis
- Arthritis
- Bone metastases
- Connective tissue diseases
- Osteoarthritis
- Fibromyalgia syndrome
- Neuropsychiatric disease
Due to the progressive nature of TIO, a delay in diagnosis and appropriate management may lead to severe osteomalacia and irreversible disability.5
HOW IS TIO DIAGNOSED?
Identifying TIO can involve assessing many factors, including5,6:
Including musculoskeletal pain, muscle weakness, fractures, fatigue, or a palpable, localized mass

Including medical history

Based on physical examination

Including fasting serum phosphorus, intact FGF23 levels, parathyroid hormone (PTH), active vitamin D, and alkaline phosphatase (ALP)

Including a gene test panel for mutations associated with hypophosphatemia that can help rule out a diagnosis of genetic forms of phosphorus wasting disorders
Including whole-body imaging
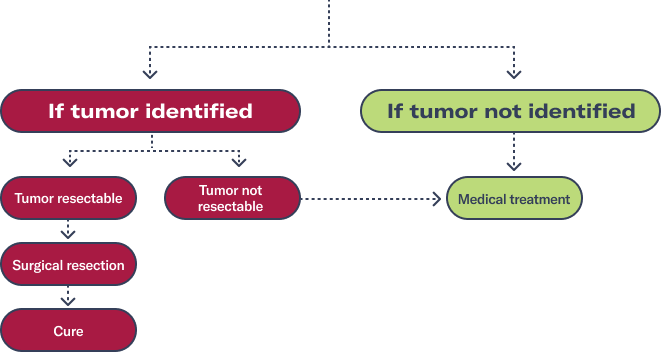
The diagnostic process for TIO6

Patient presenting with generalized musculoskeletal pain, muscle weakness, multiple fractures, fatigue, or a palpable, localized mass
- Family and medical history
- Physical examination
- Measure fasting serum phosphorus
- Biochemical workup, with or without genetic testing
- If clinical and biochemical suspicion of TIO, refer to a specialist center
- Functional and anatomical imaging
Learn how genetic and FGF23 testing can help confirm a TIO diagnosis6
Genetic testing for mutations associated with hypophosphatemia and evaluating intact FGF23 levels can help establish an accurate diagnosis for TIO.
Conduct a differential diagnosis
Learn more about sponsored genetic testinga
Check for elevated intact FGF23 levels
Request a sponsored FGF23 testing kita
aSee Terms of Use.
Common locations of TIO-causing tumors as seen
in a review of 895 patient cases7,b:
bIn a systematic clinical review of published cases that considered 5 regions for tumor localization.7
HOW IS TIO MANAGED?
Tumor resection and pharmacological intervention can help manage TIO2

Complete surgical removal of tumors that cause TIO can be curative2

When tumors cannot be localized or resected, supplements may be taken daily to help increase serum phosphorus levels2
For patients who cannot be cured by surgery, a treatment option that can help address some of the symptoms of TIO is available.2
FOR YOUR PRACTICE
Take action to help your patients

Terms of Use
Kyowa Kirin has partnered with Invitae Corp. and collaborated with Mayo Clinic Laboratories to offer sponsored, no-charge testing to patients who are being evaluated for a possible diagnosis of XLH or TIO. The Kyowa Kirin Sponsored Hypophosphatemia Program is intended to improve patient safety and quality of care by shortening the time to an accurate diagnosis, facilitate prompt confirmatory testing, and help patients with XLH and TIO meet payor coverage requirements. Use of or participation in the Kyowa Kirin Sponsored Hypophosphatemia Program does not create any obligation to use, prescribe, or recommend any Kyowa Kirin products or services. The Program is only available to patients that meet certain eligibility requirements and for whom the patient’s health care provider has determined that testing is clinically appropriate. No patient or health care provider may seek reimbursement for testing or counseling services provided under the Kyowa Kirin Sponsored Hypophosphatemia Program from any third party, including but not limited to, any government health care programs.
References:
1. Minisola S, Peacock M, Fukumoto S, et al.Tumour-induced osteomalacia. Nat Rev Dis Primers. 2017;3:17044. doi:10.1038/nrdp.2017.44 2. Dahir K, Zanchetta MB, Stanciu I, et al. Diagnosis and management of tumor-induced osteomalacia: perspectives from clinical experience. J Endocr Soc. 2021;5(9):bvab099. doi:10.1210/jendso/bvab099 3. Feng J, Jiang Y, Wang O, et al. The diagnostic dilemma of tumor induced osteomalacia: a retrospective analysis of 144 cases. Endocr J. 2017;64(7):675-683. doi:10.1507/endocrj.EJ16-0587 4. Florenzano P, Hartley IR, Jiminez M, et al. Tumor-induced osteomalacia. Calcif Tissue Int. 2021;108(1):128-142. doi:10.1007/s00223-020-00691-6 5. Brandi ML, Clunie GPR, Houillier P, et al. Challenges in the management of tumor-induced osteomalacia (TIO). Bone. 2021;152:116064. doi:10.1016/j.bone.2021.116064 6. Jan de Beur SM, Minisola S, Xia WB, et al. Global guidance for the recognition, diagnosis, and management of tumor-induced osteomalacia. J Intern Med. 2023;293(3):309-328. doi:10.1111/joim.13593 7. Bosman A, Palermo A, Vanderhulst J, et al. Tumor-induced osteomalacia: a systematic clinical review of 895 cases. Calcif Tissue Int. 2022;111(4)367-379. doi:10.1007/s00223-022-01005-8
